Microplastics in the oceans: the many ways they can harm coral life
By Cinzia Corinaldesi, Sara Canensi, Antonio DellAnno, Michael Tangherlini, Iole di Capua, Stefano Varrella, Trevor J. Willis, Carlo Cerrano and Roberto Danovaro, 15 November 2021
Submitted manuscript
Microplastics are one of the main threats to marine ecosystems, but the mechanisms determining their impact on marine life are still largely unknown. We investigated the impact of microplastics on the red coral, an emblematic and threatened species belonging to the Corallium genus, which is distributed at almost all latitudes and depths. We report here that microplastics are ingested and accumulated by corals (primarily polypropylene and polystyrene), either directly (as microplastics are confounded with the coral’s prey, plankton) or through the ingestion of zooplankton containing microplastic particles. Once ingested, microplastics cause multiple biological effects, spanning from feeding impairment and mucus release. Microplastics also cause DNA damage and a shift in the coral microbiome that together with their tissue abrasions, favour the proliferation of opportunistic/pathogenic bacteria. Since microplastic contamination is expected to double in all oceans from 2030-2060, we anticipate that their impact will likely increase in the future, with the potential of causing coral death. The effects reported on the red coral could be similar on many other habitat-forming species (species that transform the environment creating a new habitat) and may act in synergy with other stressors, potentially exacerbating the impacts of heat waves and other climate-driven events.
{1}Microplastics in the oceans: real threat to marine life?
{2}Microplastics (i.e., particles <1 mm) [N.B. Hartmann et al., 2019; M.A. Browne et al., 2015] are recognised as a potential global threat to marine life and ecosystems. These particles can be similar in size to the prey of various marine organisms [S.L. Wright et al., 2013], so that they are mistaken for their natural food and ingested, with negative effects on different forms of marine life, from the tiny zooplankton to the large fish and sharks, at all latitudes and depths down to the life inhabiting the seafloor [C.M. Rochman et al., 2016; K.L. Law, 2017; T. Kögel et al., 2019; H. Ma et al., 2020]. The huge plastic contamination of the oceans can also alter the functioning of the whole ecosystem. However, so far the biological mechanisms through which microplastics affect marine life remain largely unknown. Microplastic concentrations are not homogeneous in all seawaters, and can vary widely in marine ecosystems. Thousands of particles per m3 of seawater have been found in some coastal marine areas [N.N. Phuong et al., 2016; H.S. Auta et al., 2017; I. Paul-Pont et al., 2018] and current estimates indicate that these concentrations can be several orders of magnitude higher [J.A. Brandon et al., 2020] and are predicted to double by 2030 [A. Isobe et al., 2019]. In addition, the negative effects of microplastics are exacerbated by numerous other human impacts, including climate change.
{3}The red coral (Corallium rubrum) is one of the most known, emblematic and threatened species of the world. The genus Corallium is an ecosystem engineer, distributed at almost all latitudes and depths, from Antarctica to the equatorial latitudes and is widespread in the Mediterranean Sea [A.W. Bruckner, 2009; A.W. Bruckner, 2014; N.E. Ardila et al., 2012]. These characteristics and the very long life span (more than 200 years) make C. rubrum an ideal model to investigate the effects of microplastics on marine life and habitat forming species.
{4}How did we investigate the impacts of microplastics?
{5}We exposed red corals to different levels of microplastic contamination, spanning from those present in some contaminated coastal areas to those predicted for some seas and oceans in the next decades (2030-2060; [A. Isobe et al., 2019]). We used the same microplastic polymer composition reported from the marine environment, including polypropylene and polystyrene, which are used to produce disposable plastics (i.e., containers, bottles, cups, pipes; [I. Paul-Pont et al., 2018]) in order to simulate the natural conditions.
{6}We conducted a wide array of analyses to investigate the microplastic impact on coral feeding, defence against stress, tissue integrity (due to the physical contact with these particles), molecular response (i.e., gene expression and DNA damage), and the effects on the coral-associated microbiome (i.e., bacteria living in symbiosis with coral epidermia). We also investigated the accumulation of microplastics by the red corals, and the microplastic transfer from the prey to the predators (i.e., small crustacean larvae falling in the same size range of microplastics).
{7}The sneaky taste of the plastics: stimulating hunger while short-circuiting all biological mechanisms
{8}While corals unexposed to microplastics continued to feed without any sign of stress, the feeding activity of the corals exposed to microplastics decreased significantly with increasing microplastic concentration. After an initial stimulation of coral feeding activity, since microplastics contain phagostimulants (i.e. substances enhancing appetite), feeding activity declined and almost completely ceased at high microplastic concentrations. This response was the result of a sort of food congestion due to microplastic particles ingested [M. de Oliveira Soares et al., 2020]. At the end of the experiment, hundreds of microplastic particles (particularly the polypropylene) accumulated within each branch of red corals exposed to high microplastic concentrations. The ingestion of microplastics (particularly polystyrene) was also observed for crustacean larvae which are preyed upon by red corals, and by many other consumers/predators of the oceans.
{9}These findings indicate that microplastics can be accumulated either as a result of the direct (through direct feeding) and indirect (through predation on plankton) ingestion processes, which increase the risk of microplastic accumulation and contamination to the highest trophic levels.
{10}The dangerous touch of the microplastics
{11}After one week of the experiment, more than 20% of the coral tissue was damaged, and more than 50% of coral tissue was furrowed after two weeks, with profound lacerations which reached also the coral skeleton (figure 1). These abrasions can cause tissue necrosis [J. Reichert et al., 2018]. Regeneration mechanisms can rapidly repair small lesions of coral tissue, but healing time increases proportionately to the extent of the lesions [R. van Woesik, 1998]. In mass mortality events, tissue necrosis (with large portions of naked skeleton) have been documented in red corals and other species [C. Cerrano et al., 2000; J. Garrabou et al., 2001; J. Garrabou et al., 2009]. The mechanical abrasions observed in our study could contribute to the start of necrosis.
{12}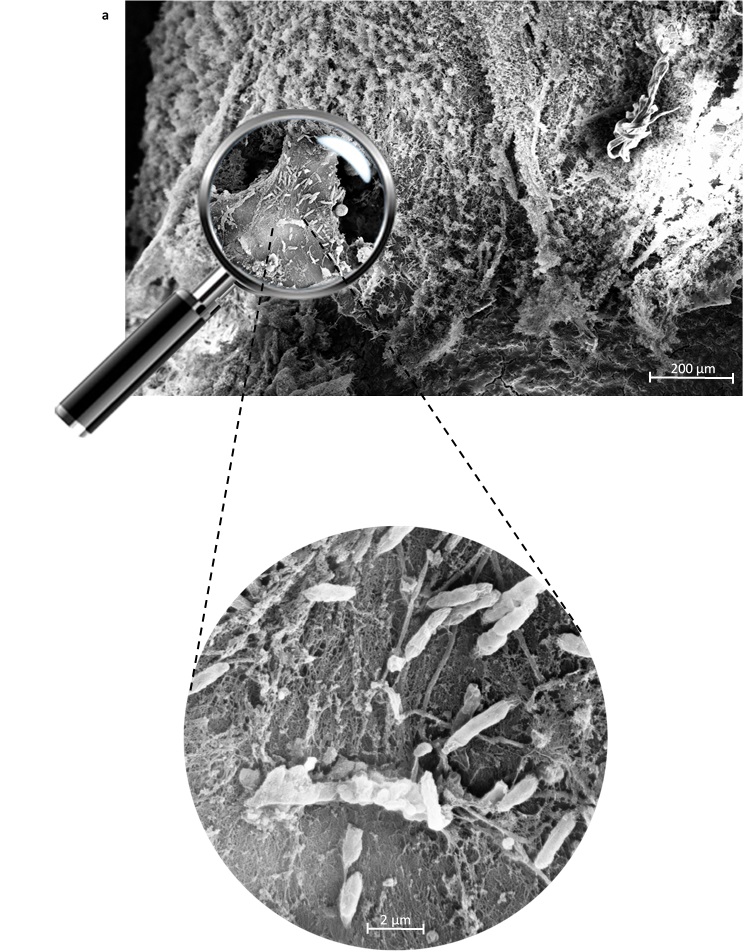
Figure 1. Damage of the red coral tissue caused by microplastic contamination. a) Tissue lacerations observed by Scanning Microscopy Analysis (SEM), and b) close-up of the coral tissue with lesions densely colonized by bacteria.
{13}From a simple “flu” to the coral DNA damage
{14}The first visible negative effect of microplastics on Carallium rubrum was the release of huge amounts of mucus, just like our flu, which is used by corals to create a protective barrier against adverse environmental conditions [B. Glasl et al., 2016; J. Reichert et al., 2018; C. Martin et al., 2019; J.A. van de Water et al., 2018]. Mucus produced by the red corals entrapped the microplastic particles along with bacteria, with amounts increasing with the increase of particle concentrations (figure 2). As a result, in a few days, the health status of the red corals worsened as demonstrated by the oxidative DNA damage and alteration of the expression levels of genes crucial to guarantee the coral defence system against stress and toxic compounds [Y.D. Louis et al., 2017; A.M. Tarrant et al., 2014].
{15}
Figure 2. Red coral (Corallium rubrum) exposed to the highest microplastic concentration enwrapped in mucus. Photo courtesy of Ettore Moretti.
{16}Microplastics affect the coral microbiomes and skin
{17}Microplastic contamination caused the increase of bacterial contamination and coral tissues were densely colonized by large microbial cells, which surrounded the lesions and induced a shift of the coral skin microbiome [J.A. van de Water et al., 2018]. The bacteria of the family Spirochaetaceae and Endozoicomonadaceae, which are typically associated with healthy red corals [J.A.J.M. van de Water et al., 2016; J.A. van de Water et al., 2018], strongly decreased or even disappeared in the corals impacted by microplastic contamination [J.A.J.M. van de Water et al., 2016; M.J. Neave et al., 2016]. Conversely, opportunistic bacteria of the families Rhodobacteraceae, Alteromanadaceae and Oceanospirillaceae increased, colonized mucus and compromised coral tissues [J.A. van de Water et al., 2018; D. Meron et al., 2011; R.L. Maher et al., 2019; M. La Riviere et al., 2013]. Some of the bacteria colonizing corals exposed to microplastics grow on these particles [E.R. Zettler et al., 2013] and contribute to degrade plastic polymers [T. Morohoshi et al., 2018]. Thus microplastics can favour the growth and/or transmission of opportunistic bacteria, which can affect the corals under physiological stress.
{18}Re-building the multiple biological effects affecting the red coral’s health
{19}The impairment of the feeding activity caused by microplastics, combined with the tissue abrasion caused by the contact with microplastics, increased the stress of C. rubrum, also atthe molecular level, causing the collapse of the cell defence, resulting in the proliferation of opportunistic bacteria and, consequently, triggering coral diseases (figure 3). Based on our findings we anticipate that all these factors combined can have cascading effects on red corals, thus causing their mortality at the concentration present in highly contaminated marine areas [Elifantz et al. 2013; Meron et al. 2011] and predicted for 2030-2060 in the oceans [A. Isobe et al., 2019].
{20}Since microplastic concentrations can be high at all depths and accumulate on the deep seafloor [K. Pabortsava and R.S. Lampitt, 2020; R. de la Fuente et al., 2021], we cannot exclude that microplastics are already causing negative biological effects on deep-water corals, similar to those reported here for the red corals and can apply to a wide range of marine life forms that share similar characteristics (e.g., filter feeders, predators and plankton feeders [H.S. Auta et al., 2017; M. de Oliveira Soares et al., 2020; M.E. Miller et al., 2020]).
{21}Microplastic contamination needs to be stopped as it has the potential to threaten red corals and other species, and such an impact could act synergistically with climate change, exacerbating the effects and frequency of the mass mortality events reported in several marine ecosystems [C. Cerrano et al., 2000].
{22}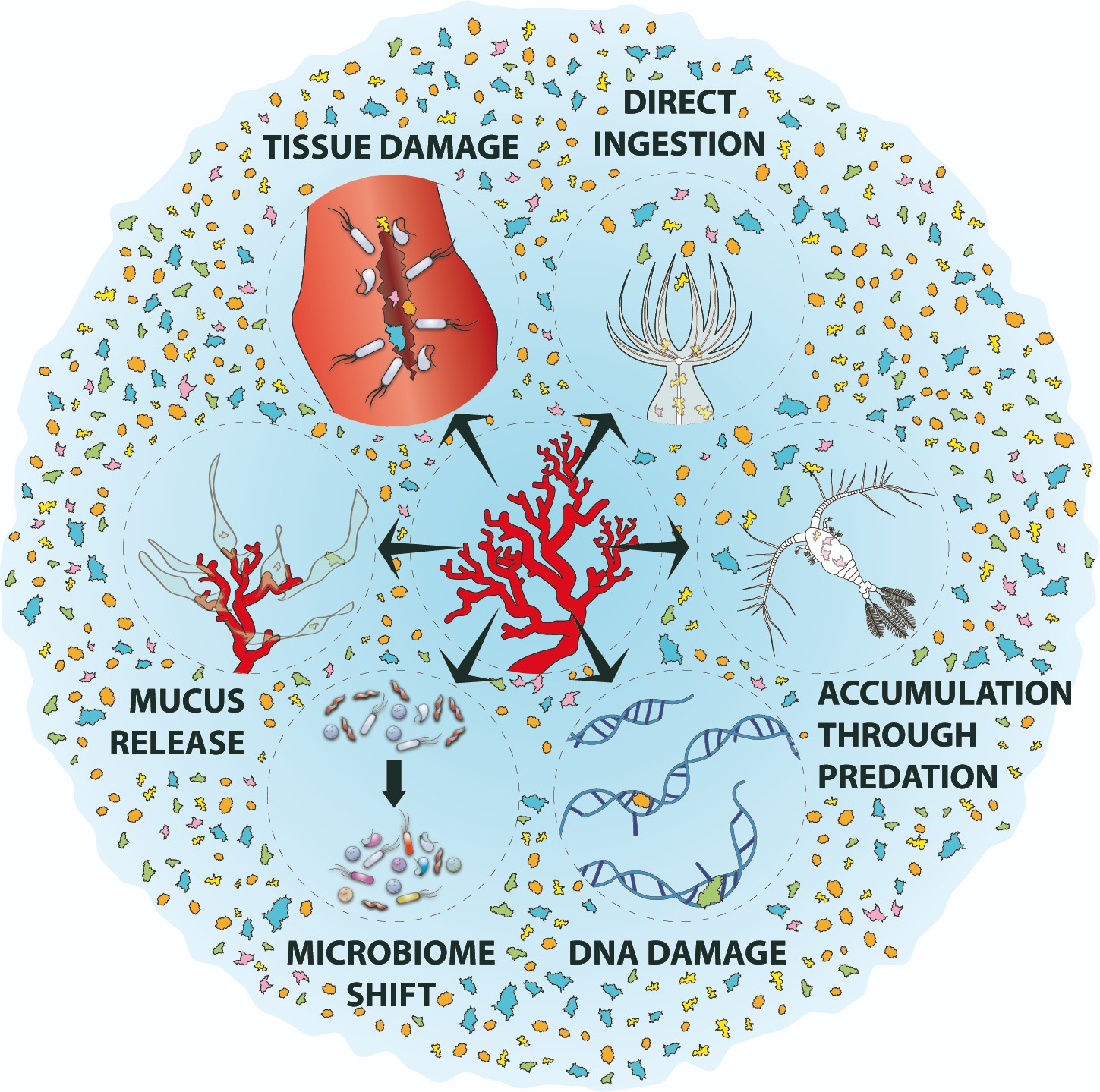
Figure 3. Conceptual model of the multiple biological and physical impacts of microplastics on the red coral. Microplastics can: accumulate within corals through direct ingestion or indirectly through predation, physically damage coral tissue, cause stress as demonstrated by the enormous release of mucus, affect DNA integrity, and cause displacement of the natural microbiome of the coral epidermis. The combination of all of these processes can ultimately cause coral mortality.
- {25}N.E. Ardila, G. Giribet and J.A. Sanchez: A time-calibrated molecular phylogeny of the precious corals: reconciling discrepancies in the taxonomic classification and insights into their evolutionary history, BMC Evol. Biol., vol. 12, 1–12, https://doi.org/10.1186/1471-2148-12-246, 2012.
- {26}H.S. Auta, C.U. Emenike and S.H. Fauziah: Distribution and importance of microplastics in the marine environment: a review of the sources, fate, effects, and potential solutions, Environ. Int., vol. 102, 165–176, https://doi.org/10.1016/j.envint.2017.02.013, 2017.
- {27}J.A. Brandon, A. Freibott and L.M. Sala: Patterns of suspended and salp ingested microplastic debris in the North Pacific investigated with epifluorescence microscopy, Limn. Oc. Lett., vol. 5, 46–53, https://doi.org/10.1002/lol2.10127, 2020.
- {28}M.A. Browne, A.J. Underwood, M.G. Chapman, R. Williams, R.C. Thompson and J.A. van Franeker: Linking effects of anthropogenic debris to ecological impacts, Proc. R. Soc. B., vol. 282, https://doi.org/10.1098/rspb.2014.2929, 2015.
- {29}A.W. Bruckner: Rate and extent of decline in Corallium (pink and red coral populations: existing data meet the requirements for a CITES Appendix II listing, Mar. Ecol. Prog. Ser., vol. 397, 319–332, https://doi.org/10.3354/meps08110, 2009.
- {30}A.W. Bruckner: Advances in management of precious corals in the family Corallidae: are new measures adequate?, Curr. Opin. Environ. Sustain., vol. 7, 1–8, https://doi.org/10.1016/j.cosust.2013.11.024, 2014.
- {31}C. Cerrano, G. Bavestrello, C.N. Bianchi, R. Cattaneo-vietti, S. Bava, C. Morganti, C. Morri, P. Picco, G. Sara and co-authors: A catastrophic mass‐mortality episode of gorgonians and other organisms in the Ligurian Sea (North‐western Mediterranean), summer 1999, Ecol. Lett., vol. 3, 284–293, https://doi.org/10.1046/j.1461-0248.2000.00152.x, 2000.
- {32}H. Elifantz, G. Horn, M. Ayon, Y. Cohen and D. Minz: Rhodobacteraceae are the key members of the microbial community of the initial biofilm formed in Eastern Mediterranean coastal seawater, FEMS Microbiol. Ecol., vol. 85, 348–357, https://doi.org/10.1111/1574-6941.12122, 2013.
- {33}R. de la Fuente, G. Drótos, E. Hernández García, C. López and E. van Sebille: Sinking microplastics in the water column: simulations in the Mediterranean Sea, Sea. Ocean Sci., vol. 17, 431–453, https://doi.org/10.5194/os-17-431-2021, 2021.
- {34}J. Garrabou, R. Coma, N. Bensoussan, M. Bally, P. Chevaldonné, M. Cigliano, D. Diaz, J.G. Harmelin, M.C. Gambi and co-authors: Mass mortality in Northwestern Mediterranean rocky benthic communities: effects of the 2003 heat wave, Glob. Change Biol., vol. 15, 1090–1103, https://doi.org/10.1111/j.1365-2486.2008.01823.x, 2009.
- {35}J. Garrabou, T. Perez, S. Sartoretto and J.G. Harmelin: Mass mortality event in red coral Corallium rubrum populations in the Provence region (France, NW Mediterranean), Mar. Ecol. Prog. Ser., vol. 217, 263–272, https://doi.org/10.3354/meps217263, 2001.
- {36}B. Glasl, G.J. Herndl, Frade and P.R.: The microbiome of coral surface mucus has a key role in mediating holobiont health and survival upon disturbance, ISME J., vol. 10, 2280, https://doi.org/10.1038/ismej.2016.9, 2016.
- {37}N.B. Hartmann, T. Hüffer, R.C. Thompson, M. Hassellöv, A. Verschoor, A.E. Daugaard, S. Rist, T. Karlsson, N. Brennholt and co-authors: Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris, Environ. Sci. Technol., vol. 53, 1039–1047, https://doi.org/10.1021/acs.est.8b05297, 2019.
- {38}A. Isobe, S. Iwasaki, K. Uchida and T. Tokai: Abundance of non-conservative microplastics in the upper ocean from 1957 to 2066, Nat. Commun., vol. 10, 1–13, https://doi.org/10.1038/s41467-019-08316-9, 2019.
- {39}T. Kögel, Ø. Bjorøy, B. Toto, A.M. Bienfait and M. Sanden: Micro-and nanoplastic toxicity on aquatic life: determining factors, Sci. Tot. Environ., vol. 709, 136050, https://doi.org/10.1016/j.scitotenv.2019.136050, 2019.
- {40}K.L. Law: Plastics in the marine environment, Annu. Rev. Mar. Sci., vol. 9, 205–229, https://doi.org/10.1146/annurev-marine-010816-060409, 2017.
- {41}Y.D. Louis, R. Bhagooli, C.D. Kenkel, A.C. Baker and S.D. Dyall: Gene expression biomarkers of heat stress in scleractinian corals: promises and limitations, Comp. Biochem. Physiol. C. Toxicol. Pharm., vol. 191, 63–77, https://doi.org/10.1016/j.cbpc.2016.08.007, 2017.
- {42}H. Ma, S. Pu, S. Liu, Y. Bai, S. Mandal and B. Xing: Microplastics in aquatic environments: toxicity to trigger ecological consequences, Environ. Pollut., vol. 261, 114089, https://doi.org/10.1016/j.envpol.2020.114089, 2020.
- {43}R.L. Maher, M.M. Rice, R. McMinds and D.E. Burkepile: Multiple stressors interact primarily through antagonism to drive changes in the coral microbiome, Sci. Rep., vol. 9, 1–12, https://doi.org/10.1038/s41598-019-43274-8, 2019.
- {44}C. Martin, E. Corona, G.A. Mahadik and C.M. Duarte: Adhesion to coral surface as a potential sink for marine microplastics, Environ. Pollut., vol. 255, 113281, https://doi.org/10.1016/j.envpol.2019.113281, 2019.
- {45}D. Meron, E. Atias, L.I. Kruh, H. Elifantz, D. Minz, M. Fine and E. Banin: The impact of reduced pH on the microbial community of the coral Acropora eurystoma, ISME J., vol. 5, 51, https://doi.org/10.1038/ismej.2010.102, 2011.
- {46}M.E. Miller, M. Hamann and F.J. Kroon: Bioaccumulation and biomagnification of microplastics in marine organisms: a review and metaanalysis of current data, PLoS ONE, vol. 15, e0240792, https://doi.org/10.1371/journal.pone.0240792, 2020.
- {47}T. Morohoshi, K. Ogata, T. Okura and S. Sato: Molecular characterization of the bacterial community in biofilms for degradation of poly (3- hydroxybutyrate-co-3-hydroxyhexanoate) films in seawater, Microbes Environ., vol. 33, 19–25, https://doi.org/10.1264/jsme2.ME17052, 2018.
- {48}M.J. Neave, A. Apprill, C. Ferrier-Pagès and C.R. Voolstra: Diversity and function of prevalent symbiotic marine bacteria in the genus Endozoicomonas, Appl. Microbiol. Biotechnol., vol. 100, 8315–8324, https://doi.org/10.1007/s00253-016-7777-0, 2016.
- {49}K. Pabortsava and R.S. Lampitt: High concentrations of plastic hidden beneath the surface of the Atlantic Ocean, Nat. Commun., vol. 11, 1–11, https://doi.org/10.1038/s41467-020-17932-9, 2020.
- {50}I. Paul-Pont, K. Tallec, C. Gonzalez-Fernandez, C. Lambert, D. Vincent, D. Mazurais, J.-L. Zambonino-Infante, G. Brotons, F. Lagarde and co-authors: Constraints and priorities for conducting experimental exposures of marine organisms to microplastics, Front. Mar. Sci., vol. 5, 252, https://doi.org/10.3389/fmars.2018.00252, 2018.
- {51}N.N. Phuong, A. Zalouk-Vergnoux, L. Poirier, A. Kamari, A. Châtel, C. Mouneyrac and F. Lagarde: Is there any consistency between the microplastics found in the field and those used in laboratory experiments?, Environ. Pollut., vol. 211, 111–123, https://doi.org/10.1016/j.envpol.2015.12.035, 2016.
- {52}J. Reichert, J. Schellenberg, P. Schubert and T. Wilke: Responses of reef building corals to microplastic exposure, Environ. Pollut., vol. 237, 955–960, https://doi.org/10.1016/j.envpol.2017.11.006, 2018.
- {53}M. La Riviere, M. Roumagnac, J. Garrabou and M. Bally: Transient shifts in bacterial communities associated with the temperate gorgonian Paramuricea clavata in the Northwestern Mediterranean Sea, PLoS ONE, vol. 8, e57385, https://doi.org/10.1371/journal.pone.0057385, 2013.
- {54}C.M. Rochman, M.A. Browne, A.J. Underwood, J.A. van Franeker, R.C. Thompson and L.A. Amaral-Zettler: The ecological impacts of marine debris: unraveling the demonstrated evidence from what is perceived, Ecology, vol. 97, 302–312, https://doi.org/10.1890/14-2070.1, 2016.
- {55}M. de Oliveira Soares, E. Matos, C. Lucas, L. Rizzo, L. Allcock and S. Rossi: Microplastics in corals: an emergent threat, Mar. Pollut. Bull., vol. 161, 111810, https://doi.org/10.1016/j.marpolbul.2020.111810, 2020.
- {56}A.M. Tarrant, A.M. Reitzel, C.K. Kwok and M.J. Jenny: Activation of the cnidarian oxidative stress response by ultraviolet radiation, polycyclic aromatic hydrocarbons and crude oil, J. Exp. Biol., vol. 217, 1444–14536, https://doi.org/10.1242/jeb.093690, 2014.
- {57}J.A. van de Water, D. Allemand and C. Ferrier-Pagès: Host-microbe interactions in octocoral holobionts-recent advances and perspectives, Microbiome, vol. 6, 64, https://doi.org/10.1186/s40168-018-0431-6, 2018.
- {58}J.A.J.M. van de Water, R. Melkonian, H. Junca, C.R. Voolstra, S. Reynaud, D. Allemand and C. Ferrier-Pagès: Spirochaetes dominate the microbial community associated with the red coral Corallium rubrum on a broad geographic scale, Sci. Rep., vol. 6, 27277, https://doi.org/10.1038/srep27277, 2016.
- {59}R. van Woesik: Lesion healing on massive Porites spp. corals, Mar. Ecol. Prog. Ser., vol. 164, 213–220, https://doi.org/10.3354/meps164213, 1998.
- {60}S.L. Wright, R.C. Thompson and T.S. Galloway: The physical impacts of microplastics on marine organisms: a review, Environ. Pollut., vol. 178, 483–492, https://doi.org/10.1016/j.envpol.2013.02.031, 2013.
- {61}E.R. Zettler, T.J. Mincer and L. Amaral-Zettler: A life in the “plastisphere”: microbial communities on plastic marine debris, Environ. Sci. Technol., vol. 47, 7137–7146, https://doi.org/10.1021/es401288x, 2013.
